More Information
Submitted: November 08, 2022 | Approved: November 23, 2022 | Published: November 24, 2022
How to cite this article: Mohammed A, Kumar P, Paranji N, Sritharan. Waghray N, Jejunal adenocarcinoma, a rare cancer of the gastrointestinal tract: a comprehensive review discussion epidemiology. Ann Clin Gastroenterol Hepatol. 2022; 6: 039-043.
DOI: 10.29328/journal.acgh.1001037
Copyright License: © 2022 Mohammed A, et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Jejunal adenocarcinoma, a rare cancer of the gastrointestinal tract: a comprehensive review discussion epidemiology
Abdul Mohammed1*, Prabhat Kumar2, Neethi Paranji1, Nithya Sritharan3 and Nisheet Waghray4
1Department of Gastroenterology and Hepatology, AdventHealth, Orlando, FL, USA
2Department of Internal Medicine, Cleveland Clinic Fairview Hospital, Cleveland OH, 44111, USA
3Department of Hematology & Oncology, Rochester General Hospital, 1425 Portland Ave, Rochester, NY, 14621, USA
4Division of Gastroenterology and Hepatology, Metro Health Medical center, Case Western Reserve University, 2500 Metrohealth Drive, Cleveland, OH, 44109, USA
*Address for Correspondence: Abdul Mohammed, MD, Department of Gastroenterology and Hepatology, AdventHealth, Orlando, Florida, United States, Email: [email protected]
Malignancy of the small bowel is exceedingly rare, accounting for 3% - 5% of all gastrointestinal malignancies [1-3]. However, in recent years because of improved diagnostic accuracy, the incidence of small bowel cancer is rising. It is estimated that 10, 470 new cases of primary SI cancer will be diagnosed in the US with 1450 cancer-related deaths [4]. The most frequent histologic types of small bowel malignant tumors include adenocarcinomas, carcinoids, lymphomas, and sarcomas. Adenocarcinoma of the small intestine is the second most common histologic type of SI cancer. The most frequent location of SI adenocarcinoma is the duodenum (57%), followed by the jejunum (29%) and ileum (13%) [5]. SI cancers are more common in men than women [6]. They occur more commonly in the African-American population and after 60 years of age [7].
There are several hypotheses to explain the relatively low incidence of SI cancers. Unlike the large intestine, a rapid transit time in the small intestine decreases exposure to luminal toxins and carcinogens. The presence of the enzyme benzopyrene hydroxylase in the intestinal mucosa aids in the detoxification and production of fewer reactive oxygen radicals in the small intestine. Further, bacterial enzymes also have oncogenic potential. A relatively low prevalence of bacteria in the small intestine is postulated to protect against carcinogenesis. The small intestine has one of the largest reserves of lymphoid tissue that confers immune surveillance against neoplastic cells. Finally, the rapid turnover of cells in the intestine is instrumental in clearing apoptotic bodies that have tumorigenic potential [8].
Risk factors
Lifestyle factors: Alcohol consumption, cigarette smoking, and dietary factors including a low-fiber diet and increased intake of processed meat and high-fructose-containing drinks are associated with increased odds of small bowel adenocarcinoma [9]. Alcohol use is associated with other gastrointestinal cancers of the esophagus, colon, and rectum [10]. It can interfere with DNA methylation, which influences cancer growth [11]. Since ethanol can also act as an irritant to the intestinal mucosa, it can increase susceptibility to carcinogens [12]. It has also been shown that acetaldehyde, the primary metabolite of ethanol, is genotoxic. In addition to alcohol, carcinogenesis with tobacco use occurs via several mechanisms. It can lead to the deposition of nitrosamines in the small intestine, a reduced cellular immune response, and impaired induction of enzymes that detoxify polycyclic aromatic hydrocarbons [13]. The resultant accumulation of reactive oxygen species predisposes cancer.
Familial syndromes
Several hereditary cancer syndromes can predispose to developing SI adenocarcinomas. Familial adenomatous polyposis (FAP) is a pre-cancerous condition associated with an increased risk of duodenal and periampullary neoplasms and early colorectal carcinoma requiring periodic surveillance. It is an autosomal dominant condition characterized by a germline mutation in the APC gene, located on chromosome 5q21 [14]. APC is a member of the Wnt/B-catenin signaling pathway that is frequently implicated in colorectal cancer. When compared with the general population, patients with FAP are associated with an elevated relative risk (RR) of duodenal adenocarcinoma (RR, 331; 95% CI 132-681) [15]. Ruys, et al. described 3 cases of jejunal adenocarcinoma that developed in patients with FAP and advanced duodenal adenomatosis [16]. The same group later performed a prospective enteroscopic evaluation in 13 patients with FAP and advanced duodenal polyposis (Spielman stage IV). Only one patient was reported to have large polyps covering one-third of the jejunal circumference and the group concluded that clinically significant jejunal polyposis was rare and routine jejunal evaluation in FAP patients was not warranted [17]. Lynch syndrome or Hereditary nonpolyposis colorectal cancer (HNPCC), an autosomal dominant disease, occurs due to germline mutations in DNA mismatch repair (MMR) genes that can predispose to not only cancers of the small intestine but also colorectal, endometrial, gastric, ovarian, biliary, and skin [18]. It is associated with a 4% lifetime risk of developing small bowel neoplasia, which predominantly involves the distal small bowel [19]. Deficiency of the MMR gene results in microsatellite instability (MSI). The National Comprehensive Cancer Network (NCCN) endorses universal MMR or MSI testing of all patients with a personal history of SI adenocarcinoma to identify individuals with Lynch syndrome. Peutz-Jeghers syndrome (PJS) is an autosomal dominant condition caused by an inherited mutation of STK11. It is characterized by multiple hamartomatous and adenomatous gastrointestinal polyps, predominantly located in the jejunum and ileum [20,21]. At-risk individuals have a relative risk of 520 for developing SI adenocarcinoma when compared with unaffected individuals [22]. The lifetime risk of SI adenocarcinoma has been estimated between 1.7% and 13% for individuals with PJS [23,24].
Crohn’s disease and celiac disease
Proinflammatory conditions like Crohn’s disease and celiac disease can also predispose to SI adenocarcinoma through an adenoma-carcinoma sequence [25,26]. The risk of adenocarcinoma of the small intestine was modestly increased (10-fold) in a large population-based study of patients with celiac disease in Sweden [27]; however, the association with the celiac disease remains poorly understood. Crohn’s disease is associated with adenocarcinoma of the distal small bowel, particularly the ileum. Von Roon, et al. described the relative risk of small intestinal cancer as 28.4 in patients with Crohn’s disease, with a mean duration of nine years prior to the development of carcinoma [28]. There are case reports of an association between proinflammatory conditions and jejunal adenocarcinoma but no large-scale RCTs have been performed [29,30].
Pathology and staging
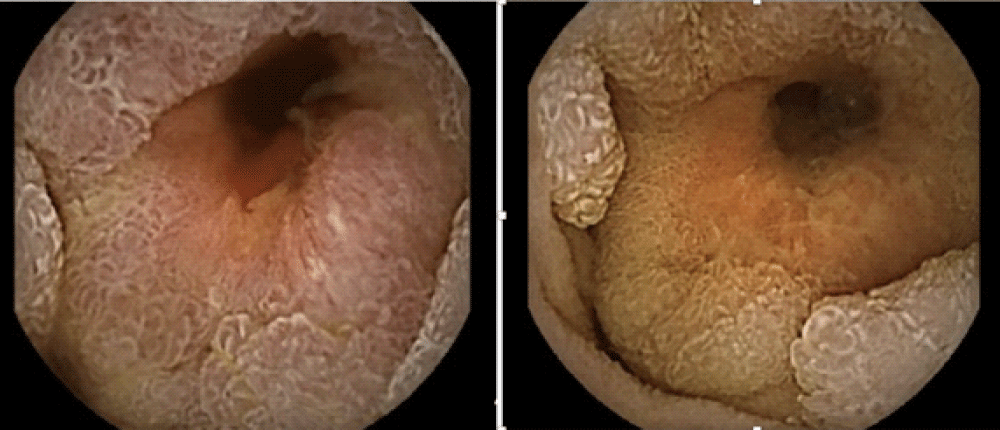
SI adenocarcinomas grossly appear as stenosing, ulcerative, infiltrative, or polypoid lesions (Figures 1,2). The histopathologic evaluation shows well to poorly differentiated tumors with a variable degree of mucin secretion. The American Joint Committee on Cancer (AJCC) stages SI adenocarcinoma in accordance with tumor size (T), regional lymphadenopathy (N), and the presence or absence of metastasis (M). Based on the extent of the disease, they are classified into 5 stages with tumor staging having a significant impact on survival; 72 months for stage I and stage II, 30 months for stage III, and 9 months for stage IV adenocarcinoma. The AJCC reported 5-year survival rates of 55% for stage I, 49% for stage IIA, 35% for stage IIB, 31% for stage IIIA, 18% for stage IIIB, and 5% for stage IV tumors [31].
Figure 1: Video capsule endoscopy demonstrating circumferential mass lesion in the proximal jejunum.
Figure 2: Push enteroscopy showing a circumferential friable mass in the proximal jejunum.
Clinical presentation and diagnosis
Bridge, et al. described the clinical and pathological features of 32 adenocarcinomas of the jejunum [32]. Patients with jejunal adenocarcinoma may be asymptomatic or have non-specific symptoms such as intermittent abdominal cramping or pain, nausea, vomiting, or weight loss. Based on the size, location, and blood supply, adenocarcinomas of the jejunum can present with intestinal obstruction or occult gastrointestinal bleeding. At the time of diagnosis, more patients with jejunal adenocarcinoma are reported to be symptomatic when compared to duodenal adenocarcinoma (84% vs. 57%) [33]. Because of non-specific symptoms, there is a significant delay in diagnosing disease averaging 7 to 8 months from the onset of symptoms [34].
Several diagnostic modalities are available to evaluate small intestinal lesions. Barium studies are not particularly effective in identifying distal small intestinal lesions with a sensitivity of 50% [35]. In contrast, Computed tomography (CT) enterography and Magnetic resonance (MR) enterography have the advantage of providing multiplanar cross-sectional imaging over enteroclysis with a sensitivity of 100%. A prospective study comparing CT enterography to MR enterography in 150 patients with suspected small bowel disease reported that MR enterography was more accurate than CT enterography, particularly for neoplastic diseases (p = .0412) [36]. Video capsule endoscopy (VCE) and enteroscopy are the preferred diagnostic modalities to provide direct visualization of jejunal lesions. Zhang, et al. reported a higher detection rate of SI tumors with DBE when compared to VCE [37]. VCE allows complete visualization of the small bowel mucosa and is particularly effective in diagnosing lesions in patients with obscure GI bleeding with a detection rate of 4% to 9% [38]. In the case of small bowel obstruction or stricture, enteroscopy is the preferred diagnostic technique owing to the risk of capsule retention.
Molecular mechanisms
Genetic alterations in SI adenocarcinomas are not very well documented. K-ras mutations were more common in jejunal adenocarcinoma (43% to 47%), while p - 53 mutations are more common in duodenal adenocarcinoma (41% to 48%) [39]. While ras genes regulate intracellular signaling pathways by encoding guanine nucleotide-binding proteins, K-ras activation has demonstrated oncogenic potential. Inactivation of p -53 tumor suppression genes and resultant deletion of chromosome 17p results in dysregulation of apoptosis. A mutation in the APC gene, a component of the Wnt/ B-catenin signaling pathway, that is frequently linked with colorectal cancer is seen as increasingly associated with periampullary tumors and duodenal tumors. Microsatellite instability arising from a deficiency in the MMR gene in Lynch syndrome can be found in 5% to 35% of SI adenocarcinomas, predominantly affecting the distal small bowel [40].
Treatment
There are no prospective randomized trials that have evaluated surgery for the treatment of primary SI adenocarcinoma but retrospective reviews have been reported. The type of surgical resection used to treat localized cancer depends on the location of the tumor. Surgical resection with en-bloc removal of regional lymph nodes offers the best chance of overall survival in patients with local (stage I-III) jejunal adenocarcinoma. The rate of curative resection (R0) is 64% to 97% with a median survival time of 19 months [41,42]. The 5-year cancer-specific survival rates were 66% for jejunal lesions [43]. After surgical resection, local and distant recurrences are common.
The role and efficacy of chemotherapy in various stages of SI adenocarcinoma remain unclear. The phase III BALLAD trial is the first prospective study evaluating the role of adjuvant 5-fluorouracil/leucovorin/oxaliplatin (FOLFOX) compared with observation alone for patients with stage I-III disease [44]. The results of the BALLAD trial are yet to be reported, meanwhile, data from retrospective studies are limited. There are no phase 3 clinical trials assessing the efficacy of chemotherapy in unresectable and stage IV jejunal adenocarcinoma. Retrospective studies with a 5-fluorouracil/leucovorin/oxaliplatin (FOLFOX4) regimen report an overall survival of 20 months for stage III disease and 12 months for stage IV jejunal adenocarcinoma [45]. Currently, the NCCN recommends systemic therapy for advanced SI adenocarcinoma. There are three chemotherapy regimens: FOLFOX, capecitabine plus oxaliplatin (CAPEOX), or 5-fluorouracil, leucovorin, oxaliplatin, irinotecan (FOLFOXIRI), any of which may be combined with bevacizumab for patients with advanced disease who are appropriate for intensive therapy. For patients who are not appropriate for intensive therapy, treatment options would include 5-fluorouracil and leucovorin or capecitabine with or without bevacizumab [46].
Prognosis
Overall survival in SI adenocarcinoma is significantly correlated with tumor location, staging, age of the patient, surgical resection, and history of Crohn’s disease [47]. The most important independent prognostic factors on a multivariate analysis include curative resection (R0), lymph node involvement, and the ratio of positive to negative lymph nodes [48]. In patients undergoing curative resection, a poor prognosis was associated with patients who are older than 55 years of age, African-American heritage, duodenal or distal or diffuse tumor localization, advanced TNM stage, metastatic, poorly differentiated, or involved margins [49]. Several studies suggest that jejunal tumors have a better prognosis than duodenal tumors [1,41].
In conclusion, adenocarcinoma of jejunum is a rare cause of iron deficiency anemia. Considering its non-specific presenting symptoms, a high index of suspicion should be maintained, particularly when an EGD and colonoscopy do not exhibit the source of bleeding. Risk factors include environmental, familial, and chronic inflammatory conditions. Curative surgical resection is the treatment of choice for localized cancer, while systemic chemotherapy is reserved for advanced disease.
- Dabaja BS, Suki D, Pro B, Bonnen M, Ajani J. Adenocarcinoma of the small bowel: presentation, prognostic factors, and outcome of 217 patients. Cancer. 2004 Aug 1;101(3):518-26. doi: 10.1002/cncr.20404. PMID: 15274064.
- Cancer of the Small Intestine - Cancer Stat Facts. SEER. [cited 2021 Mar 3]. Available from: https://seer.cancer.gov/statfacts/html/smint.html
- Aparicio T, Zaanan A, Svrcek M, Laurent-Puig P, Carrere N, Manfredi S, Locher C, Afchain P. Small bowel adenocarcinoma: epidemiology, risk factors, diagnosis and treatment. Dig Liver Dis. 2014 Feb;46(2):97-104. doi: 10.1016/j.dld.2013.04.013. Epub 2013 Jun 21. PMID: 23796552.
- Siegel RL, Miller KD, Jemal A. Cancer statistics, 2018. CA Cancer J Clin. 2018 Jan;68(1):7-30. doi: 10.3322/caac.21442. Epub 2018 Jan 4. PMID: 29313949.
- Halfdanarson TR, McWilliams RR, Donohue JH, Quevedo JF. A single-institution experience with 491 cases of small bowel adenocarcinoma. Am J Surg. 2010 Jun;199(6):797-803. doi: 10.1016/j.amjsurg.2009.05.037. PMID: 20609724.
- Haselkorn T, Whittemore AS, Lilienfeld DE. Incidence of small bowel cancer in the United States and worldwide: geographic, temporal, and racial differences. Cancer Causes Control. 2005 Sep;16(7):781-7. doi: 10.1007/s10552-005-3635-6. PMID: 16132788.
- Aparicio T, Henriques J, Manfredi S, Tougeron D, Bouché O, Pezet D, Piessen G, Coriat R, Zaanan A, Legoux JL, Terrebone E, Pocard M, Gornet JM, Lecomte T, Lombard-Bohas C, Perrier H, Lecaille C, Lavau-Denes S, Vernerey D, Afchain P; NADEGE Investigators. Small bowel adenocarcinoma: Results from a nationwide prospective ARCAD-NADEGE cohort study of 347 patients. Int J Cancer. 2020 Aug 15;147(4):967-977. doi: 10.1002/ijc.32860. Epub 2020 Jan 22. PMID: 31912484.
- Varghese R, Weedon R. 'Metachronous' adenocarcinoma of the small intestine. Int J Clin Pract Suppl. 2005 Apr;(147):106-8. doi: 10.1111/j.1368-504x.2005.00070.x. PMID: 15875642.
- Bennett CM, Coleman HG, Veal PG, Cantwell MM, Lau CC, Murray LJ. Lifestyle factors and small intestine adenocarcinoma risk: A systematic review and meta-analysis. Cancer Epidemiol. 2015 Jun;39(3):265-73. doi: 10.1016/j.canep.2015.02.001. Epub 2015 Feb 27. PMID: 257368603.
- Wiseman M. The second World Cancer Research Fund/American Institute for Cancer Research expert report. Food, nutrition, physical activity, and the prevention of cancer: a global perspective. Proc Nutr Soc. 2008 Aug;67(3):253-6. doi: 10.1017/S002966510800712X. Epub 2008 May 1. PMID: 18452640.
- Varela-Rey M, Woodhoo A, Martinez-Chantar ML, Mato JM, Lu SC. Alcohol, DNA methylation, and cancer. Alcohol Res. 2013;35(1):25-35. PMID: 24313162; PMCID: PMC3860423.
- Burbige EJ, Lewis DR Jr, Halsted CH. Alcohol and the gastrointestinal tract. Med Clin North Am. 1984 Jan;68(1):77-89. doi: 10.1016/s0025-7125(16)31242-1. PMID: 6361419.
- IARC Working Group on the Evaluation of Carcinogenic Risks to Humans. Tobacco smoke and involuntary smoking. IARC Monogr Eval Carcinog Risks Hum. 2004;83:1-1438. PMID: 15285078; PMCID: PMC4781536.
- Galiatsatos P, Foulkes WD. Familial adenomatous polyposis. Am J Gastroenterol. 2006 Feb;101(2):385-98. doi: 10.1111/j.1572-0241.2006.00375.x. PMID: 16454848.
- Offerhaus GJ, Giardiello FM, Krush AJ, Booker SV, Tersmette AC, Kelley NC, Hamilton SR. The risk of upper gastrointestinal cancer in familial adenomatous polyposis. Gastroenterology. 1992 Jun;102(6):1980-2. doi: 10.1016/0016-5085(92)90322-p. PMID: 1316858.
- Ruys AT, Alderlieste YA, Gouma DJ, Dekker E, Mathus-Vliegen EM. Jejunal cancer in patients with familial adenomatous polyposis. Clin Gastroenterol Hepatol. 2010 Aug;8(8):731-3. doi: 10.1016/j.cgh.2010.04.008. Epub 2010 Apr 24. PMID: 20399906.
- Alderlieste YA, Rauws EA, Mathus-Vliegen EM, Fockens P, Dekker E. Prospective enteroscopic evaluation of jejunal polyposis in patients with familial adenomatous polyposis and advanced duodenal polyposis. Fam Cancer. 2013 Mar;12(1):51-6. doi: 10.1007/s10689-012-9571-1. PMID: 23054214.
- Bonadona V, Bonaïti B, Olschwang S, Grandjouan S, Huiart L, Longy M, Guimbaud R, Buecher B, Bignon YJ, Caron O, Colas C, Noguès C, Lejeune-Dumoulin S, Olivier-Faivre L, Polycarpe-Osaer F, Nguyen TD, Desseigne F, Saurin JC, Berthet P, Leroux D, Duffour J, Manouvrier S, Frébourg T, Sobol H, Lasset C, Bonaïti-Pellié C; French Cancer Genetics Network. Cancer risks associated with germline mutations in MLH1, MSH2, and MSH6 genes in Lynch syndrome. JAMA. 2011 Jun 8;305(22):2304-10. doi: 10.1001/jama.2011.743. PMID: 21642682.
- Haanstra JF, Al-Toma A, Dekker E, Vanhoutvin SA, Nagengast FM, Mathus-Vliegen EM, van Leerdam ME, de Vos tot Nederveen Cappel WH, Sanduleanu S, Veenendaal RA, Cats A, Vasen HF, Kleibeuker JH, Koornstra JJ. Prevalence of small-bowel neoplasia in Lynch syndrome assessed by video capsule endoscopy. Gut. 2015 Oct;64(10):1578-83. doi: 10.1136/gutjnl-2014-307348. Epub 2014 Sep 10. PMID: 25209657.
- Shibata HR, Phillips MJ. Peutz-Jeghers syndrome with jejunal and colonic adenocarcinomas. Can Med Assoc J. 1970 Aug 1;103(3):285-7. PMID: 4317531; PMCID: PMC1930385.
- Matuchansky C, Babin P, Coutrot S, Druart F, Barbier J, Maire P. Peutz-Jeghers syndrome with metastasizing carcinoma arising from a jejunal hamartoma. Gastroenterology. 1979 Dec;77(6):1311-5. PMID: 499717.
- Giardiello FM, Brensinger JD, Tersmette AC, Goodman SN, Petersen GM, Booker SV, Cruz-Correa M, Offerhaus JA. Very high risk of cancer in familial Peutz-Jeghers syndrome. Gastroenterology. 2000 Dec;119(6):1447-53. doi: 10.1053/gast.2000.20228. PMID: 11113065.
- Shenoy S. Genetic risks and familial associations of small bowel carcinoma. World J Gastrointest Oncol. 2016 Jun 15;8(6):509-19. doi: 10.4251/wjgo.v8.i6.509. PMID: 27326320; PMCID: PMC4909452.
- Hearle N, Schumacher V, Menko FH, Olschwang S, Boardman LA, Gille JJ, Keller JJ, Westerman AM, Scott RJ, Lim W, Trimbath JD, Giardiello FM, Gruber SB, Offerhaus GJ, de Rooij FW, Wilson JH, Hansmann A, Möslein G, Royer-Pokora B, Vogel T, Phillips RK, Spigelman AD, Houlston RS. Frequency and spectrum of cancers in the Peutz-Jeghers syndrome. Clin Cancer Res. 2006 May 15;12(10):3209-15. doi: 10.1158/1078-0432.CCR-06-0083. PMID: 16707622.
- Rampertab SD, Forde KA, Green PH. Small bowel neoplasia in coeliac disease. Gut. 2003 Aug;52(8):1211-4. doi: 10.1136/gut.52.8.1211. PMID: 12865284; PMCID: PMC1773745.
- Caio G, Volta U, Ursini F, Manfredini R. Small bowel adenocarcinoma as a complication of celiac disease: clinical and diagnostic features. BMC Gastroenterol . 2019 Mar 27 [cited 2021 Feb 13];19. Available from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6437995/
- Askling J, Linet M, Gridley G, Halstensen TS, Ekström K, Ekbom A. Cancer incidence in a population-based cohort of individuals hospitalized with celiac disease or dermatitis herpetiformis. Gastroenterology. 2002 Nov;123(5):1428-35. doi: 10.1053/gast.2002.36585. PMID: 12404215.
- von Roon AC, Reese G, Teare J, Constantinides V, Darzi AW, Tekkis PP. The risk of cancer in patients with Crohn's disease. Dis Colon Rectum. 2007 Jun;50(6):839-55. doi: 10.1007/s10350-006-0848-z. PMID: 17308939.
- Ursi P, Tarallo M, Crocetti D, Cavallaro G, Fiori E, D'Andrea V, De Toma G. Second jejunal loop adenocarcinoma associated with celiac disease: the first case report. G Chir. 2019 May-Jun;40(3):225-229. PMID: 31484013.
- Vecchio R, Marchese S, Gangemi P, Alongi G, Ferla F, Spataro C, Intagliata E. Laparoscopic treatment of mucinous adenocarcinoma of jejunum associated with celiac disease. Case report. G Chir. 2012 Apr;33(4):126-8. PMID: 22668531.
- Amin MB, Edge S, Greene F. editors. AJCC Cancer Staging Manual [Internet]. 8th ed. Springer International Publishing. 2017; [cited 2020 Nov 6]. Available from: https://www.springer.com/gp/book/9783319406176
- Bridge MF, Perzin KH. Primary adenocarcinoma of the jejunum and ileum. A clinicopathologic study. Cancer. 1975 Nov;36(5):1876-87. doi: 10.1002/1097-0142(197511)36:5<1876::aid-cncr2820360545>3.0.co;2-7. PMID: 53095.
- Sakae H, Kanzaki H, Nasu J, Akimoto Y, Matsueda K, Yoshioka M, Nakagawa M, Hori S, Inoue M, Inaba T, Imagawa A, Takatani M, Takenaka R, Suzuki S, Fujiwara T, Okada H. The characteristics and outcomes of small bowel adenocarcinoma: a multicentre retrospective observational study. Br J Cancer. 2017 Nov 21;117(11):1607-1613. doi: 10.1038/bjc.2017.338. Epub 2017 Oct 5. PMID: 28982111; PMCID: PMC5729438.
- Ciresi DL, Scholten DJ. The continuing clinical dilemma of primary tumors of the small intestine. Am Surg. 1995 Aug;61(8):698-702; discussion 702-3. PMID: 7618809.
- Mata A, Llach J, Castells A, Rovira JM, Pellisé M, Ginès A, Fernández-Esparrach G, Andreu M, Bordas JM, Piqué JM. A prospective trial comparing wireless capsule endoscopy and barium contrast series for small-bowel surveillance in hereditary GI polyposis syndromes. Gastrointest Endosc. 2005 May;61(6):721-5. doi: 10.1016/s0016-5107(05)00289-0. PMID: 15855978.
- Masselli G, Di Tola M, Casciani E, Polettini E, Laghi F, Monti R, Bernieri MG, Gualdi G. Diagnosis of Small-Bowel Diseases: Prospective Comparison of Multi-Detector Row CT Enterography with MR Enterography. Radiology. 2016 May;279(2):420-31. doi: 10.1148/radiol.2015150263. Epub 2015 Nov 24. PMID: 26599801.
- Zhang ZH, Qiu CH, Li Y. Different roles of capsule endoscopy and double-balloon enteroscopy in obscure small intestinal diseases. World J Gastroenterol. 2015 Jun 21;21(23):7297-304. doi: 10.3748/wjg.v21.i23.7297. PMID: 26109818; PMCID: PMC4476893.
- Cobrin GM, Pittman RH, Lewis BS. Increased diagnostic yield of small bowel tumors with capsule endoscopy. Cancer. 2006 Jul 1;107(1):22-7. doi: 10.1002/cncr.21975. PMID: 16736516.
- Rashid A, Hamilton SR. Genetic alterations in sporadic and Crohn's-associated adenocarcinomas of the small intestine. Gastroenterology. 1997 Jul;113(1):127-35. doi: 10.1016/s0016-5085(97)70087-8. PMID: 9207270.
- Schrock AB, Devoe CE, McWilliams R, Sun J, Aparicio T, Stephens PJ, Ross JS, Wilson R, Miller VA, Ali SM, Overman MJ. Genomic Profiling of Small-Bowel Adenocarcinoma. JAMA Oncol. 2017 Nov 1;3(11):1546-1553. doi: 10.1001/jamaoncol.2017.1051. PMID: 28617917; PMCID: PMC5710195.
- Talamonti MS, Goetz LH, Rao S, Joehl RJ. Primary cancers of the small bowel: analysis of prognostic factors and results of surgical management. Arch Surg. 2002 May;137(5):564-70; discussion 570-1. doi: 10.1001/archsurg.137.5.564. PMID: 11982470.
- Ugurlu MM, Asoglu O, Potter DD, Barnes SA, Harmsen WS, Donohue JH. Adenocarcinomas of the jejunum and ileum: a 25-year experience. J Gastrointest Surg. 2005 Nov;9(8):1182-8. doi: 10.1016/j.gassur.2005.08.027. PMID: 16269390.
- Wilhelm A, Galata C, Beutner U, Schmied BM, Warschkow R, Steffen T, Brunner W, Post S, Marti L. Duodenal localization is a negative predictor of survival after small bowel adenocarcinoma resection: A population-based, propensity score-matched analysis. J Surg Oncol. 2018 Mar;117(3):397-408. doi: 10.1002/jso.24877. Epub 2017 Oct 16. PMID: 29044591.
- Kitahara H, Honma Y, Ueno M, Kanemitsu Y, Ohkawa S, Mizusawa J, Furuse J, Shimada Y; Colorectal Cancer Study Group and Hepatobiliary and Pancreatic Oncology Group of the Japan Clinical Oncology Group. Randomized phase III trial of post-operative chemotherapy for patients with stage I/II/III small bowel adenocarcinoma (JCOG1502C, J-BALLAD). Jpn J Clin Oncol. 2019 Mar 1;49(3):287-290. doi: 10.1093/jjco/hyy188. PMID: 30590606.
- Chen CW, Wang WM, Su YC, Wu JY, Hsieh JS, Wang JY. Oxaliplatin/5-fluorouracil/leucovorin (FOLFOX4) regimen as an adjuvant chemotherapy in the treatment of advanced jejunal adenocarcinoma: a report of 2 cases. Med Princ Pract. 2008;17(6):496-9. doi: 10.1159/000151574. Epub 2008 Oct 3. PMID: 18836281.
- Benson AB, Venook AP, Al-Hawary MM, Arain MA, Chen YJ, Ciombor KK, Cohen SA, Cooper HS, Deming DA, Garrido-Laguna I, Grem JL, Hoffe SE, Hubbard J, Hunt S, Kamel A, Kirilcuk N, Krishnamurthi S, Messersmith WA, Meyerhardt J, Miller ED, Mulcahy MF, Nurkin S, Overman MJ, Parikh A, Patel H, Pedersen KS, Saltz LB, Schneider C, Shibata D, Skibber JM, Sofocleous CT, Stoffel EM, Stotsky-Himelfarb E, Willett CG, Johnson-Chilla A, Gregory KM, Gurski LA. Small Bowel Adenocarcinoma, Version 1.2020, NCCN Clinical Practice Guidelines in Oncology. J Natl Compr Canc Netw. 2019 Sep 1;17(9):1109-1133. doi: 10.6004/jnccn.2019.0043. PMID: 31487687.
- Legué LM, Bernards N, Gerritse SL, van Oudheusden TR, de Hingh IH, Creemers GM, Ten Tije AJ, Lemmens VE. Trends in incidence, treatment and survival of small bowel adenocarcinomas between 1999 and 2013: a population-based study in The Netherlands. Acta Oncol. 2016 Sep-Oct;55(9-10):1183-1189. doi: 10.1080/0284186X.2016.1182211. Epub 2016 May 12. PMID: 27170100.
- Wu TJ, Yeh CN, Chao TC, Jan YY, Chen MF. Prognostic factors of primary small bowel adenocarcinoma: univariate and multivariate analysis. World J Surg. 2006 Mar;30(3):391-8; discussion 399. doi: 10.1007/s00268-005-7898-6. PMID: 16479330.
- Bilimoria KY, Bentrem DJ, Wayne JD, Ko CY, Bennett CL, Talamonti MS. Small bowel cancer in the United States: changes in epidemiology, treatment, and survival over the last 20 years. Ann Surg. 2009 Jan;249(1):63-71. doi: 10.1097/SLA.0b013e31818e4641. PMID: 19106677.